The emission spectrum of a chemical element or compound is the spectrum of frequencies of electromagnetic radiation emitted when electrons transition from a higher energy state to a lower energy state. Different elements have unique spectra due to variations in the number of protons, electrons, and their arrangements, which result in differences in the amount of energy absorbed and emitted during electron movements.
These emission spectra act as fingerprints for identifying elements.
Understanding Emission Spectrum
The emission spectrum of each element in the periodic table is unique and reflects the specific frequencies of electromagnetic radiation emitted when electrons transition from higher to lower energy states. Different elements have different spectra due to variations in the number of protons and arrangement of electrons.
This creates a captivating display of colored lines, known as line or atomic spectra, which can be observed and studied.
Definition And Significance Of Emission Spectrum:
An emission spectrum refers to the spectrum of frequencies of electromagnetic radiation that is emitted when electrons transition from a higher energy state to a lower energy state. This phenomenon is crucial in understanding the behavior of elements and compounds, and it has several important implications:
- Identifying Elements: Each element has its unique emission spectrum, which can be used to identify and characterize it. By analyzing the spectrum produced by a sample, scientists can determine the presence of specific elements.
- Atomic Structure: Emission spectra provide valuable insights into the atomic structure of an element. The discrete lines observed in the spectrum correspond to the energy levels at which electrons reside within the atom.
- Energy Transitions: The emission spectrum helps us understand the energy transitions that electrons undergo. By analyzing the emitted frequencies, scientists can deduce the energy differences between various electron orbits.
- Research and Applications: Emission spectra are widely used in scientific research and practical applications. They are employed in fields such as astronomy, chemistry, and materials science to study and analyze the behavior of different substances.
The emission spectrum is a powerful tool in scientific investigations, enabling us to gain a deeper understanding of the atomic world and its behavior.
Exploring Spectral Lines
Explore the fascinating world of spectral lines in the emission spectrum periodic table. Discover how different elements emit unique frequencies of electromagnetic radiation, providing valuable insights into their composition and energy states. Uncover the secrets hidden within the colorful lines of light and delve into the wonders of emission spectroscopy.
Spectral Lines And Their Characteristics:
- Spectral lines refer to the specific wavelengths or frequencies of light that are emitted or absorbed by an atom or molecule.
- Each element or compound has a unique set of spectral lines that can be used to identify its presence.
- Spectral lines can be categorized into two types: Emission lines and absorption lines.
- Emission lines occur when electrons in an atom or molecule are excited to a higher energy state and then return to a lower energy state, releasing energy in the form of photons.
- Absorption lines occur when electrons in an atom or molecule absorb photons of specific wavelengths and transition to higher energy levels.
Relationship Between Energy Levels And Spectral Lines:
- The energy levels of electrons in an atom or molecule determine the wavelengths of light that will be emitted or absorbed.
- When an electron transitions from a higher energy level to a lower energy level, it releases energy in the form of a photon with a specific wavelength.
- The difference in energy levels between the initial and final states of an electron corresponds to the energy of the emitted or absorbed photon.
- The relationship between energy levels and spectral lines is quantized, meaning that only certain energy levels are allowed, resulting in discrete wavelengths or frequencies of light.
Quantized Nature Of Spectral Lines:
- Spectral lines are quantized because electronic energy levels in atoms and molecules are quantized.
- This means that only specific energy levels are allowed for electrons, and transitions between these levels result in specific wavelengths or frequencies of light.
- The quantized nature of spectral lines is a consequence of the wave-particle duality of electrons, where they can act as both particles and waves.
- The quantization of spectral lines is a fundamental principle of quantum mechanics, which describes the behavior of particles at the atomic and subatomic level.
- By studying the quantized nature of spectral lines, scientists can gain insights into the energy levels and properties of atoms and molecules and contribute to fields such as spectroscopy and quantum chemistry.
Why Do Elements Have Different Spectra?
Different elements have unique spectra because of the variations in the number of protons and electrons they possess, resulting in different energy levels. As a result, each element emits or absorbs specific frequencies of electromagnetic radiation, creating its distinct emission spectrum.
Different elements have different spectra because of various factors that influence the emission spectrum. Let’s explore them:
Role Of Protons And Electrons In Determining The Spectrum:
- Protons: The number of protons in an atom’s nucleus determines its atomic number and, subsequently, its identity as a specific element.
- Electrons: The arrangement of electrons around the nucleus plays a crucial role in determining an element’s spectral lines.
Energy Absorption And Emission By Atoms:
- Absorption: When atoms absorb energy, their electrons move to higher energy levels.
- Emission: As the excited electrons return to their original energy levels, they emit energy in the form of light or electromagnetic radiation.
Factors influencing energy absorption and emission include:
- Energy levels: Each electron occupies a specific energy level or shell around the nucleus.
- Transition between energy levels: When electrons transition between energy levels, they emit or absorb energy corresponding to specific wavelengths.
Elements With Unique Arrangements Of Electrons:
- Electrons occupy specific orbitals and energy levels within an atom.
- The arrangement of electrons within an element’s electron configuration determines the wavelengths of light it absorbs and emits.
Elements have different spectra due to the role of protons and electrons, energy absorption and emission by atoms, and unique arrangements of electrons. Each element’s unique properties result in distinct patterns of spectral lines, making spectroscopy a valuable tool for identifying elements.

Credit: wires.onlinelibrary.wiley.com
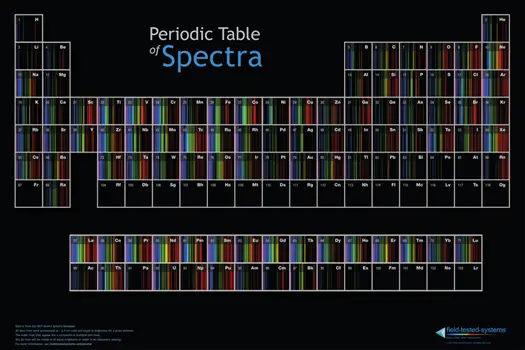
Unveiling The Periodic Table Of Spectra
Unveiling the Periodic Table of Spectra reveals the emission spectrum of each element, showcasing the unique frequencies of electromagnetic radiation emitted by their electrons as they transition from high energy to low energy states. This groundbreaking resource by Field Tested Systems in Austin, Texas brings the world of emission spectroscopy to life.
When it comes to understanding the behavior of elements and the unique characteristics they possess, scientists rely on the emission spectrum. The emission spectrum of a chemical element or compound showcases the frequencies of electromagnetic radiation that are emitted when electrons transition from a higher energy state to a lower one.
This spectrum is crucial in unraveling the secrets of elements and their behavior.
Introduction To The Periodic Table Of Spectra
The Periodic Table of Spectra serves as a valuable tool for scientists to study and analyze the emission spectrum of various elements. It arranges these elements in a structured manner, providing crucial information about their spectral characteristics. This table allows researchers to observe patterns and trends, making it easier to identify and distinguish elements based on their emission spectra.
Key factors in arranging the elements:
- Similar patterns: The table arranges elements in a way that groups elements with similar spectral patterns together, aiding in easier identification and comparison.
- Atomic number: Each element is listed in order of increasing atomic number, offering a systematic arrangement that is easy to follow.
How The Table Is Arranged
The Periodic Table of Spectra follows a strategic arrangement that provides insights into how elements emit radiation. Here’s how it is organized:
- Periods: The table consists of horizontal rows called periods, with each period representing a distinct energy level or shell of electrons.
- Groups: In addition to periods, the table is also divided into vertical columns known as groups. Elements within the same group possess similar outer electron configurations, influencing their emission spectra.
- Blocks: The table is further divided into blocks, including s, p, d, and f blocks, based on the type of subshell being filled with electrons.
The arrangement of the elements in the Periodic Table of Spectra provides scientists with a visual representation of how the emission spectrum varies across elements, allowing for better analysis and understanding.
Significance And Applications Of The Table
The Periodic Table of Spectra holds immense significance in the field of chemistry and beyond. Here are a few key applications:
- Identification of elements: By referring to the emission spectra displayed in the table, scientists can identify unknown elements based on their unique spectral patterns, allowing for easy classification.
- Light source analysis: The table aids in the analysis of different light sources by comparing their emission spectra to known elements. This helps in identifying the composition of light sources, such as stars or artificial light.
- Materials research: Understanding the emission spectra of elements is crucial in material science research. It allows scientists to develop new materials with specific optical properties by manipulating the arrangement of atoms and their energy levels.
- Forensic analysis: Emission spectroscopy techniques, based on the Periodic Table of Spectra, are utilized in forensic analysis to identify substances present at a crime scene. By comparing the emission spectra of unknown substances to the known spectra in the table, scientists can determine their composition.
The Periodic Table of Spectra plays a vital role in unraveling the mysteries of elements and their emission spectra. Its strategic arrangement and detailed information provide scientists with key insights for various applications, making it a valuable tool in the world of chemistry and beyond.
Analyzing Emission Spectra Of Representative Elements
Analyzing the emission spectra of representative elements provides valuable insights into the unique characteristics and energy transitions of each element. This information can be found in the periodic table, offering a comprehensive understanding of the emission spectrum for each element.
The emission spectrum of a chemical element is a fascinating topic to explore in the field of spectroscopy. By analyzing the emission spectra of representative elements, we can gain valuable insights into the unique characteristics of each element’s spectral pattern.
In this section, we will take a closer look at a case study: the hydrogen atom’s emission spectrum.
Case Study: Hydrogen Atom’S Emission Spectrum
The hydrogen atom holds a special place in emission spectroscopy due to its simple and well-understood structure. When excited, hydrogen atoms emit light at specific wavelengths, creating a distinct emission spectrum. Let’s explore the notable emission lines of hydrogen:
- Balmer series: This series consists of four prominent emission lines in the visible spectrum. These lines are labeled H-alpha (656.3 nm), H-beta (486.1 nm), H-gamma (434.0 nm), and H-delta (410.2 nm). Each line corresponds to a transition between different energy levels of the hydrogen atom.
- Lyman series: These emission lines occur in the ultraviolet region of the electromagnetic spectrum. The most notable line in this series is Lyman-alpha (121.6 nm), which corresponds to the transition from the second energy level to the first energy level of the hydrogen atom.
- Paschen series: This series of emission lines falls in the infrared region of the spectrum. The most significant line in this series is Paschen-alpha (1875.1 nm), representing the transition from the fourth energy level to the third energy level of the hydrogen atom.
The spectral pattern of hydrogen can be explained by the quantized nature of electrons in atoms. When excited, electrons jump from higher energy levels to lower energy levels, releasing energy in the form of photons with specific frequencies. This phenomenon gives rise to the unique emission lines observed in hydrogen’s spectrum.
Analyzing the emission spectra of representative elements, such as the hydrogen atom, provides valuable insights into the behavior of atoms and the quantum nature of energy transitions. The distinct emission lines in hydrogen’s spectrum further deepen our understanding of atomic structure and the quantized behavior of electrons.
Exploring Emission Spectra Of Other Elements
Explore the emission spectra of other elements on the periodic table to discover the unique frequencies of electromagnetic radiation emitted when electrons transition between energy states. Each element has its own distinct spectrum, reflecting the differences in energy absorption and emission.
Analysis Of Emission Spectra For Select Elements:
- Hydrogen: The emission spectrum of hydrogen is one of the most well-known and important spectra in physics. It consists of a series of lines that correspond to different transitions of electrons within the hydrogen atom. These lines, known as the Balmer series, are in the visible part of the spectrum and appear as distinct colors when viewed through a spectroscope.
- Sodium: Sodium also has a characteristic emission spectrum. It emits a bright yellow-orange color when heated. This is a result of the transitions of electrons in the sodium atom. The spectrum of sodium is commonly observed in street lamps and other sources of yellow light.
- Calcium: The emission spectrum of calcium is more complex, consisting of multiple lines in the visible spectrum. Each line corresponds to a different transition between energy levels in the calcium atom. Calcium is often used as a reference element for spectroscopic measurements due to its well-defined emission lines.
Understanding The Variations In Spectral Patterns:
- Each element has a unique emission spectrum: The emission spectrum of an element is determined by the energy levels and transitions of electrons within its atoms. As a result, each element has a distinct pattern of spectral lines that can be used to identify and analyze its presence.
- Spectral patterns can provide information about atomic structure: The positions and intensities of the spectral lines in an emission spectrum can reveal valuable insights about the electronic configuration of an element. By studying these patterns, scientists can gain a better understanding of the behavior of atoms and their interactions with electromagnetic radiation.
- Spectral patterns can be influenced by external factors: While the emission spectrum of an element is primarily determined by its atomic structure, external factors such as temperature and pressure can also affect the spectral patterns. Changes in these factors can lead to shifts or broadening of the spectral lines, providing additional information about the conditions under which the spectrum is observed.
Factors Influencing The Emission Spectra:
- Energy levels of electrons: The emission spectrum of an element is directly related to the energy levels of its electrons. Different energy transitions result in the emission of photons with specific frequencies, which manifest as spectral lines in the spectrum. The specific arrangement of energy levels in an atom determines the unique pattern of spectral lines for each element.
- Electron configurations: The distribution and arrangement of electrons in an atom’s energy levels influence the emission spectrum. Different electron configurations result in different possible transitions, leading to variations in the spectral patterns. The number of electrons in each energy level and the overall electron configuration play crucial roles in determining the emission spectrum.
- Interactions with other elements or compounds: The presence of other elements or compounds can influence the emission spectrum of an element. Interactions such as collisions or chemical reactions can alter the energy levels and electron transitions, leading to changes in the spectral patterns observed. This can be useful in studying the dynamics of chemical reactions or identifying unknown substances through their emission spectra.
By analyzing the emission spectra of various elements, scientists can unlock valuable information about atomic structure, electron behavior, and interactions with other substances. Understanding the variations in spectral patterns and the factors influencing them is crucial for the fields of spectroscopy and atomic physics.
Applications Of Emission Spectra
The application of emission spectra plays a crucial role in understanding the periodic table. By analyzing the unique frequencies of electromagnetic radiation emitted by each element, scientists can identify and study their composition and properties. This information is essential for various fields such as chemistry, astronomy, and materials science.
Emission Spectrum Periodic Table
Emission spectra play a crucial role in various fields, offering valuable insights into the composition and characteristics of elements. From identifying elements to practical applications in chemistry and physics, emission spectra have proven to be a powerful tool. In this section, we will explore the applications of emission spectra in different disciplines.
Utilization Of Emission Spectra In Various Fields:
- Chemistry: Emission spectra provide a unique “fingerprint” for each element, making it possible to identify elements present in a sample. This information is pivotal in areas such as forensic analysis, environmental monitoring, and quality control in industries.
- Astronomy: By studying the emission spectra of celestial bodies, astronomers can determine the chemical composition of stars and galaxies. This helps in understanding the formation and evolution of the universe.
- Material Science: Emission spectra aid in the characterization of materials, offering valuable insights into their atomic structure and chemical properties. This information is essential for developing new materials with specific properties for various applications.
Identification Of Elements Using Emission Spectra:
- Each element possesses a unique emission spectrum due to the transitions of electrons between energy levels. This characteristic allows scientists to identify unknown elements by comparing their emission spectra with known standards.
- The emission spectra of elements can be observed using various techniques such as flame tests, gas discharge tubes, and spectroscopy. These methods enable scientists to analyze the specific wavelengths of light emitted by elements, aiding in their identification.
Practical Applications In Chemistry And Physics:
- Drug Discovery: Emission spectra help chemists analyze the interactions between molecules and determine the structure and behavior of compounds. This information plays a crucial role in the development of new drugs and understanding their mechanism of action.
- Energy Research: Emission spectra assist researchers in studying the behavior of atoms and molecules involved in energy transfer processes. This knowledge is vital for the development of efficient energy sources such as solar cells and batteries.
- Quantum Mechanics: Emission spectra provide valuable experimental data to validate theoretical models in quantum mechanics. This field of study helps scientists understand the behavior of particles at the atomic and subatomic levels.
By harnessing the power of emission spectra, scientists and researchers can unlock a deeper understanding of the fundamental building blocks of matter. From identifying elements to advancing various scientific disciplines, emission spectra continue to be an invaluable tool in pushing the boundaries of knowledge.
Frequently Asked Questions Of Emission Spectrum Periodic Table
What Is The Emission Spectrum Of An Element?
The emission spectrum of an element is the spectrum of electromagnetic radiation emitted when electrons transition from a high energy state to a lower energy state.
Is Emission Spectrum Different For Each Element?
Each element has a different emission spectrum due to variations in the number of protons and arrangement of electrons.
What Is The Periodic Table Of Spectra?
The periodic table of spectra captures the unique spectrum of each element, which serves as a fingerprint for identification.
What Is The Line Spectrum Of Each Element?
The line spectrum of each element is a unique set of colored lines observed when atoms emit light.
Conclusion
To conclude, the emission spectrum of each element in the periodic table is unique and serves as a fingerprint for identification purposes. It is a result of electrons transitioning from high energy states to lower energy states, emitting electromagnetic radiation at specific frequencies.
The differences in spectra among elements are due to variations in the number of protons and the arrangement of electrons. When atoms are excited, they emit light at specific wavelengths, which correspond to different colors. These emitted lights form a series of colored lines, known as line or atomic spectra.
Field Tested Systems has developed a Spectra Table that captures the unique spectrum of each element, allowing for easy identification. Understanding and studying the emission spectra of elements is essential in various scientific fields. By analyzing these spectra, scientists can gain insights into the composition and properties of different elements.